|
Elements are arranged in periods and groups, each providing valuable information about their characteristics. The periodic table on the MCAT organizes all known elements based on their atomic number, representing the number of protons in an atom's nucleus. In this section, we will explore the importance of the MCAT periodic table and how a mastery of its structure and trends can enhance your performance on the exam. 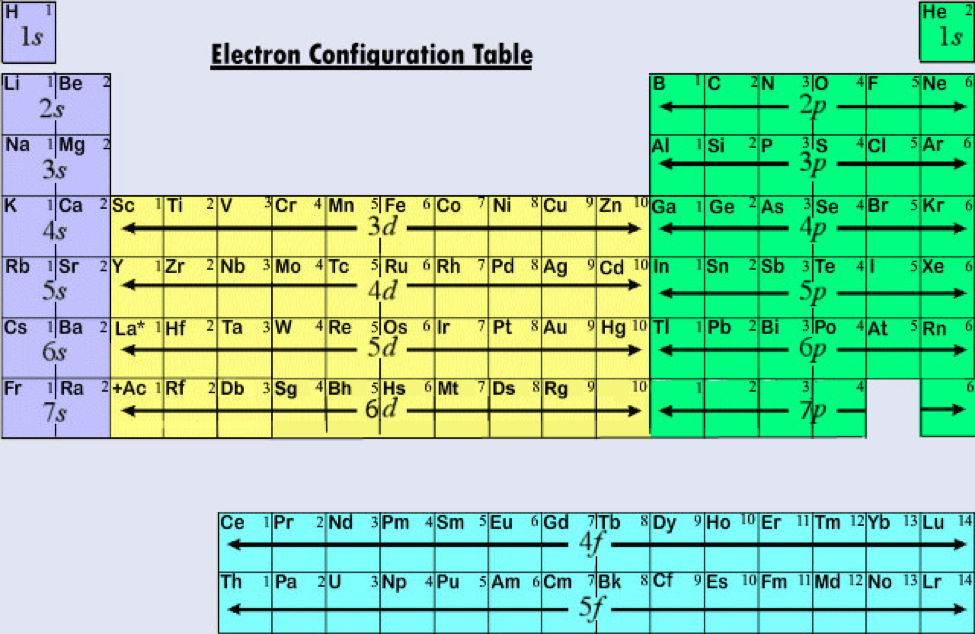 
The MCAT extensively tests your knowledge of this table and its trends, as it serves as the foundation for understanding various concepts in chemistry, biochemistry, and even biology. Gaining a comprehensive grasp of the periodic table is essential for aspiring medical students. The periodic table in the MCAT is not just a chart filled with elements and their symbols it is a fundamental tool that holds the key to understanding the behavior and properties of chemical elements. We’ll delve into and explore the significance of this foundational knowledge and provide the necessary insights to excel in this vital examination aspect. Whether you're a prospective medical student or simply seeking a deeper understanding of chemistry, this guide will equip you with the essential knowledge to confidently tackle the MCAT. 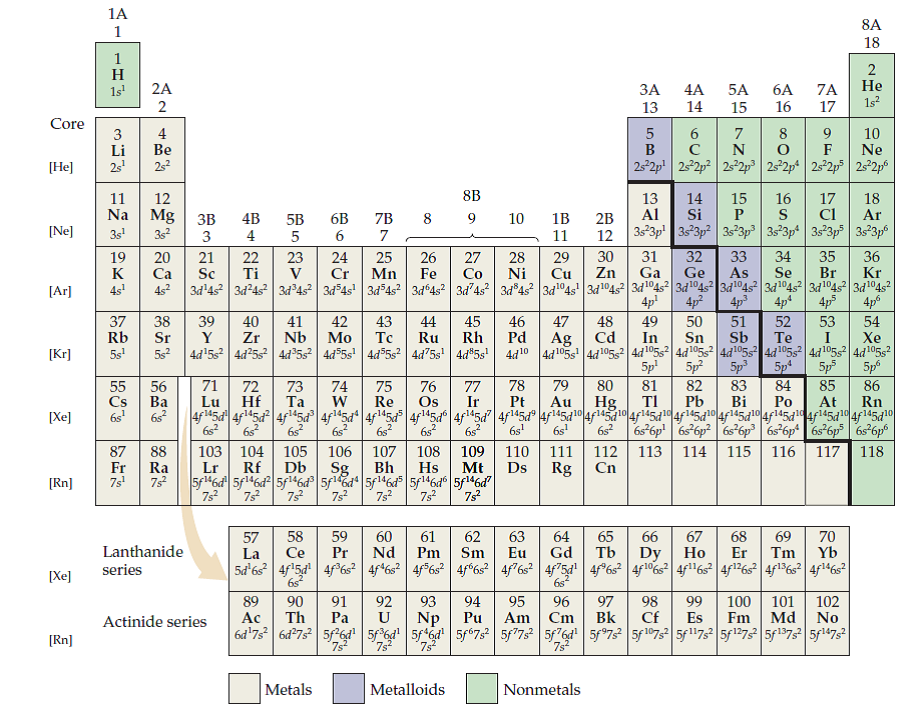
While some schools do not require the MCAT, for most students, this exam is a crucial step in their future careers. The periodic table is among the fundamental concepts tested on the MCAT, which lays the foundation for understanding chemical elements and their properties. The Medical College Admission Test ( MCAT) is a crucial gateway for aspiring medical students that assesses their knowledge and aptitude across various scientific disciplines. Former Admissions Committee Member, Hofstra-Northwell School of Medicineįamiliar with the MCAT periodic table? Read on to learn everything you need to know.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
